The Two Isoforms of Lyn Display Different Intramolecular Fuzzy Complexes with the SH3 Domain
João M. C. Teixeira, Héctor Fuentes, Stasė Bielskutė, Margarida Gairi, Szymon Żerko, Wiktor Koźmiński, Miquel Pons
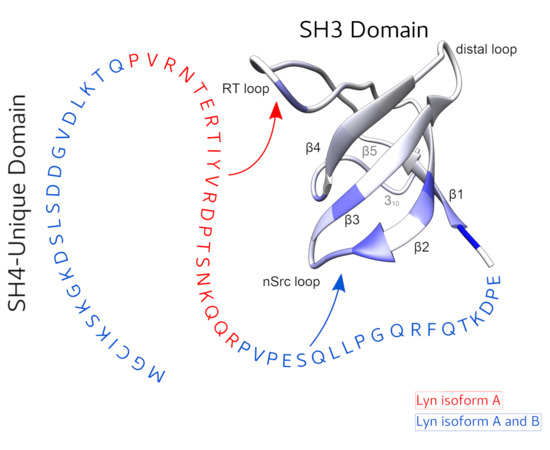
The function of the intrinsically disordered Unique domain of the Src family of tyrosine kinases (SFK), where the largest differences between family members are concentrated, remains poorly understood. Recent studies in c-Src have demonstrated that the Unique region forms transient interactions, described as an intramolecular fuzzy complex, with the SH3 domain and suggested that similar complexes could be formed by other SFKs. Src and Lyn are members of a distinct subfamily of SFKs. Lyn is a key player in the immunologic response and exists in two isoforms originating from alternative splicing in the Unique domain. We have used NMR to compare the intramolecular interactions in the two isoforms and found that the alternatively spliced segment interacts specifically with the so-called RT-loop in the SH3 domain and that this interaction is abolished when a polyproline ligand binds to the SH3 domain. These results support the generality of the fuzzy complex formation in distinct subfamilies of SFKs and its physiological role, as the naturally occurring alternative splicing modulates the interactions in this complex.
